MEDICAL DEVICE FOR THE PURPOSE OF EXPORT ONLY
MEDICAL DEVICE FOR THE PURPOSE OF EXPORT ONLY
1. Introduction
The importation, exportation, or placement of a medical device in the Malaysia market requires the medical device to be registered under Medical Device Act 2012 (Act 737). In accordance with the Medical Device (Exemption) Order 2024, the medical devices for the purpose of export only are exempted from registration requirements. The exporter who wishes to export the medical device from Malaysia shall submit an application for an exemption and shall comply with the requirements as determined by the Authority.
An “Medical Device for Export Only Exemption Letter” letter is issued by the Authority then permits the medical device to be exported.
2. Requirements on the application for an exemption of Medical Device for the purpose of Export Only
- The applicant who wishes to export the medical device from Malaysia shall submit an application for an exemption to the Authority prior to the
- One application shall be made for only one single medical device nomenclature
- The complete application together with the supporting document as per Table 1 shall be submitted via Google Form
Table 1: Document required on the application for an exemption of Medical Device for the purpose of Export Only
|
No |
Documents |
|
1 |
Registrar of Companies (ROC) certificate of applicant |
|
2 |
|
|
3 |
|
|
4 |
(Please refer to Figure 2: Determination whether LOA is required or not for Export Only Exemption) |
|
5 |
A copy manufacturer’s QMS ISO 13485 certificates |
|
6 |
A copy of Product Datasheet/Brochure/Leaflet/Label/IFU that contain information on brief description and intended use. |
|
7 |
A copy of establishment license (If applicable) |
3. Determination whether LOA is required or not for an exemption of Medical Device for the purpose of Export Only
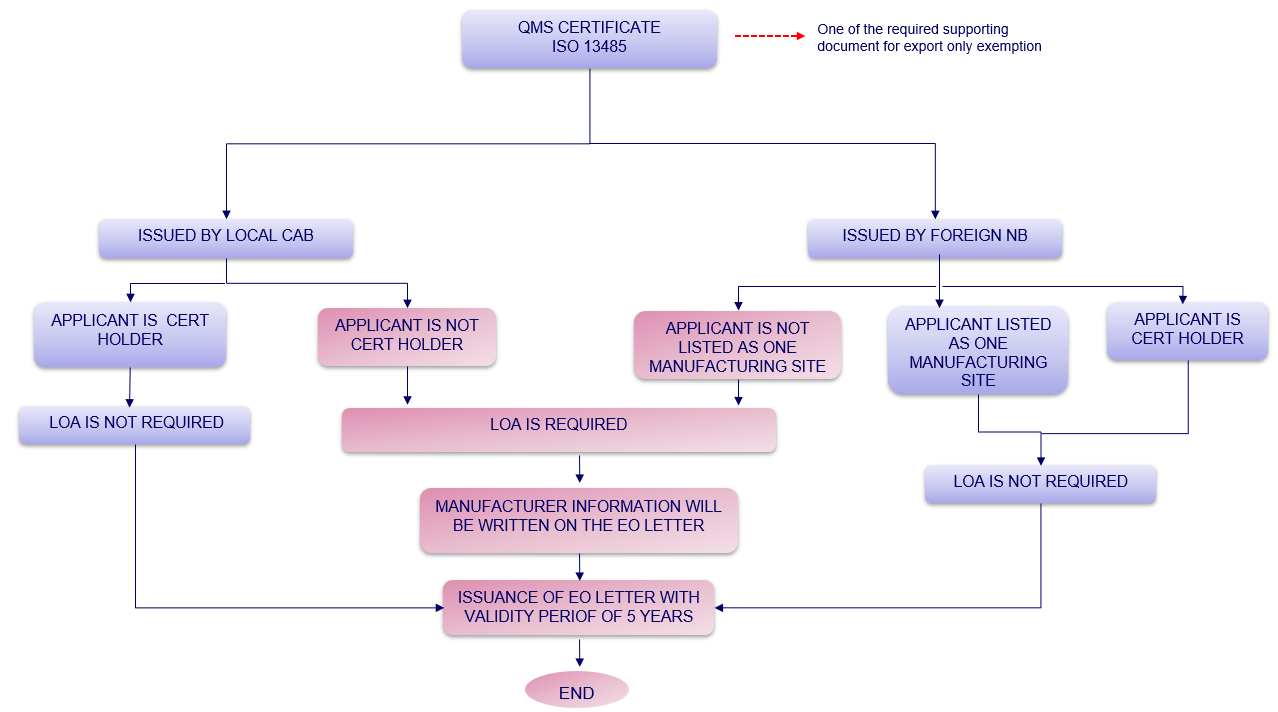
Figure 1: Determination whether LOA is required or not for Notification of Export Only Medical Device
4. Process flow the application for an exemption of medical device for the purpose of export only
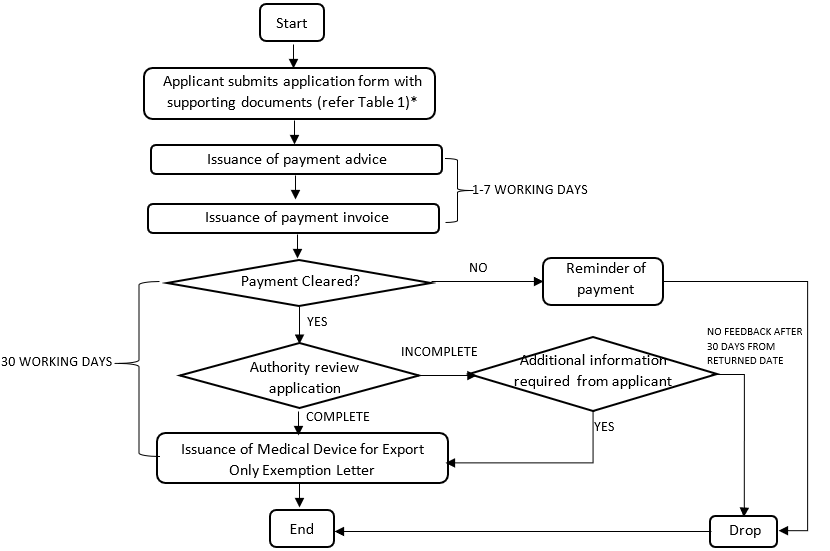
Figure 2: Process flow the application for an exemption of medical device for the purpose of export only
5. Fees for the application for an exemption of medical device for the purpose of export only
The payment RM500 shall be made online via portal BayarNow. For guidance on online payment is accessible via link or qr code below:

6. Issuance of Medical Device for Export Only Exemption Letter
- The validity period of the Medical Device for Export Only Exemption Letter is five (5) years.
- There is no renewal for the application for an exemption of Medical Device Export Only. Therefore, the applicant shall submit for a new application and the validity date as per the new application approved date.
Any inquiries please email to exportonly.ec@mda.gov.my
General Line: +603 8230 0300
Direct Line: +603 8230 0253 (Pn. Nur Aisyah)
+603 8230 0299 (En. Ziyad)
+603 8230 0208 (Pn. Hafizah)
Updated: 09 / 06 / 2026